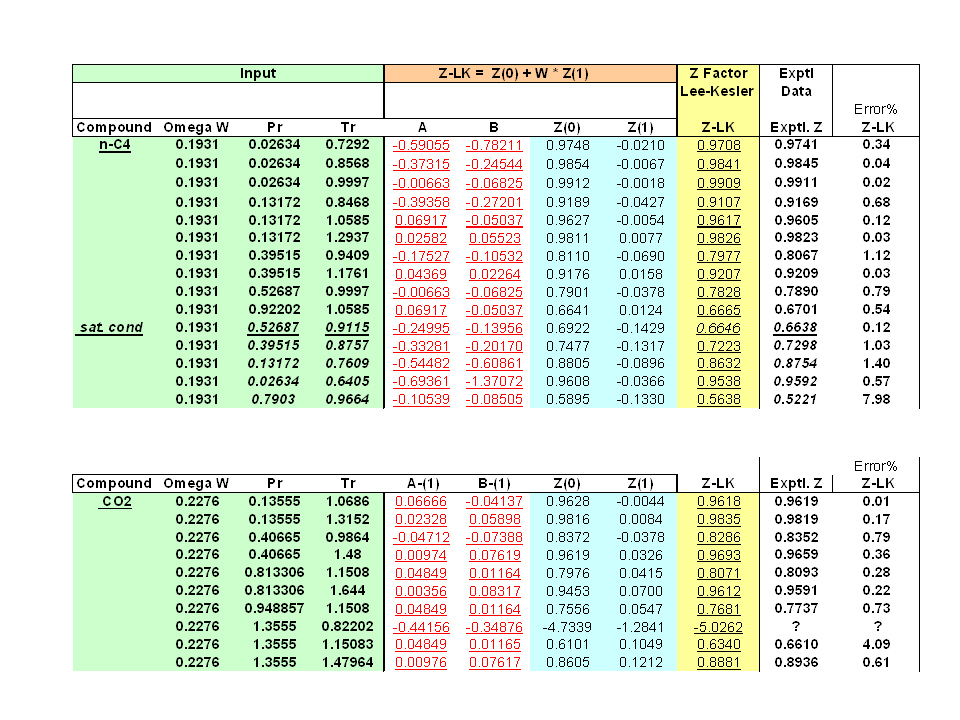
Write the expression for the compressibility factor (Z) for one

Solved We begin by showing that the compressibility factor

Determine Compressibility of Gases

SOLVED: 4.17 The non-ideality of a gas may be expressed as a compressibility factor, z: PVm RT a. Find the value of z for the ideal gas. b. Given the van der

1st PUC Chemistry Model Question Paper 5 with Answers (Old Pattern) - KSEEB Solutions

Solved RT B 2. The compressiblity factor for a gas is

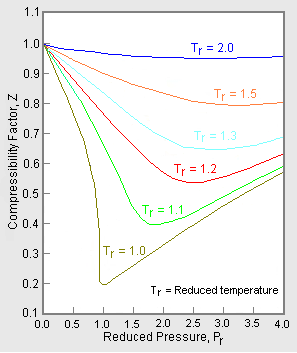
Gas compressibility factor Z: Ideal gas vs Real gas

Compressibility factor Z versus ρ ¯ for the n = 4 fluid. The plot

Bengali] The compresibility factor (Z) of one mole of a van der waals

The compressibility factor (Z) of one mole of a van der Waals' gas of negligible 'a ' value is:1dfrac{bp}{RT}1+dfrac{bp}{RT}1-dfrac{bp}{RT}
Compressibility factor (gases) - Citizendium

Compressibility Factor (Z-Factor) Equation of State

SOLVED: Derive an expression for the compression factor of a gas

1. The compressibility factor, z, is the ratio of

Functional-Segment Activity Coefficient Equation of State: F-SAC-Phi