physical chemistry - Is the compressibility factor smaller or greater than 1 at low temperature and high pressure? - Chemistry Stack Exchange
4.8
(231)
Write Review
More
$ 24.50
In stock
Description
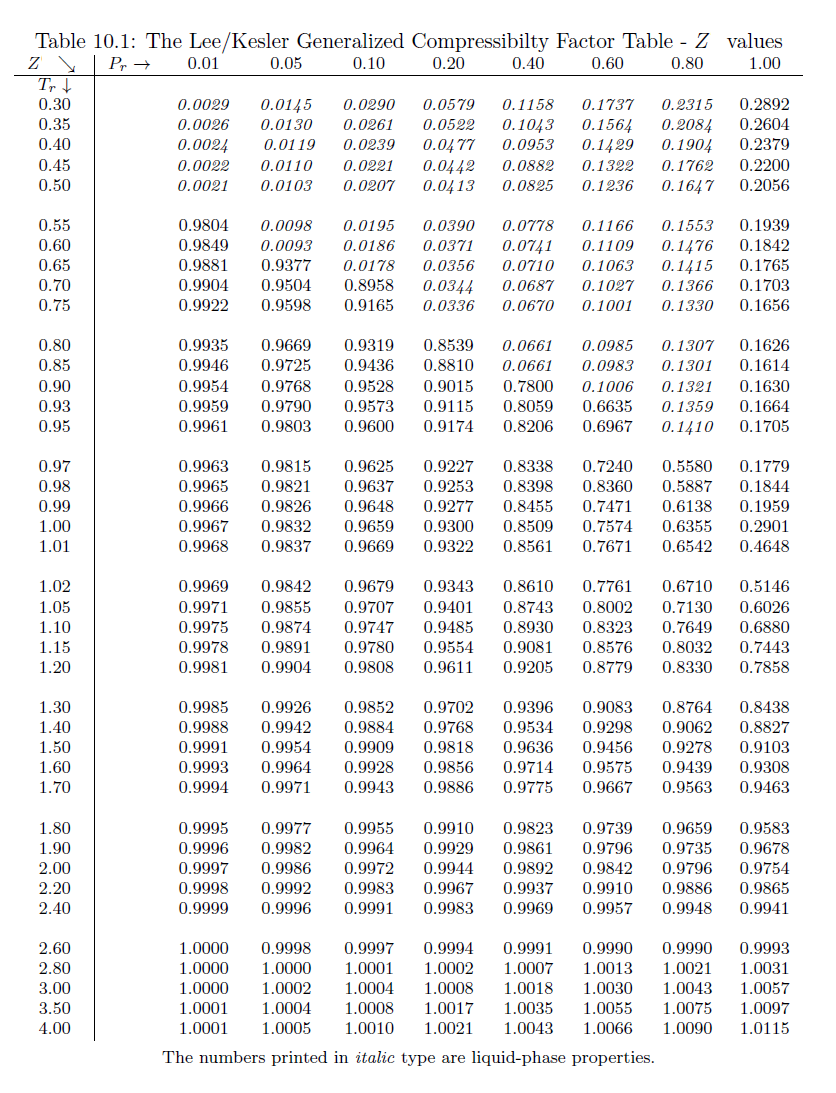
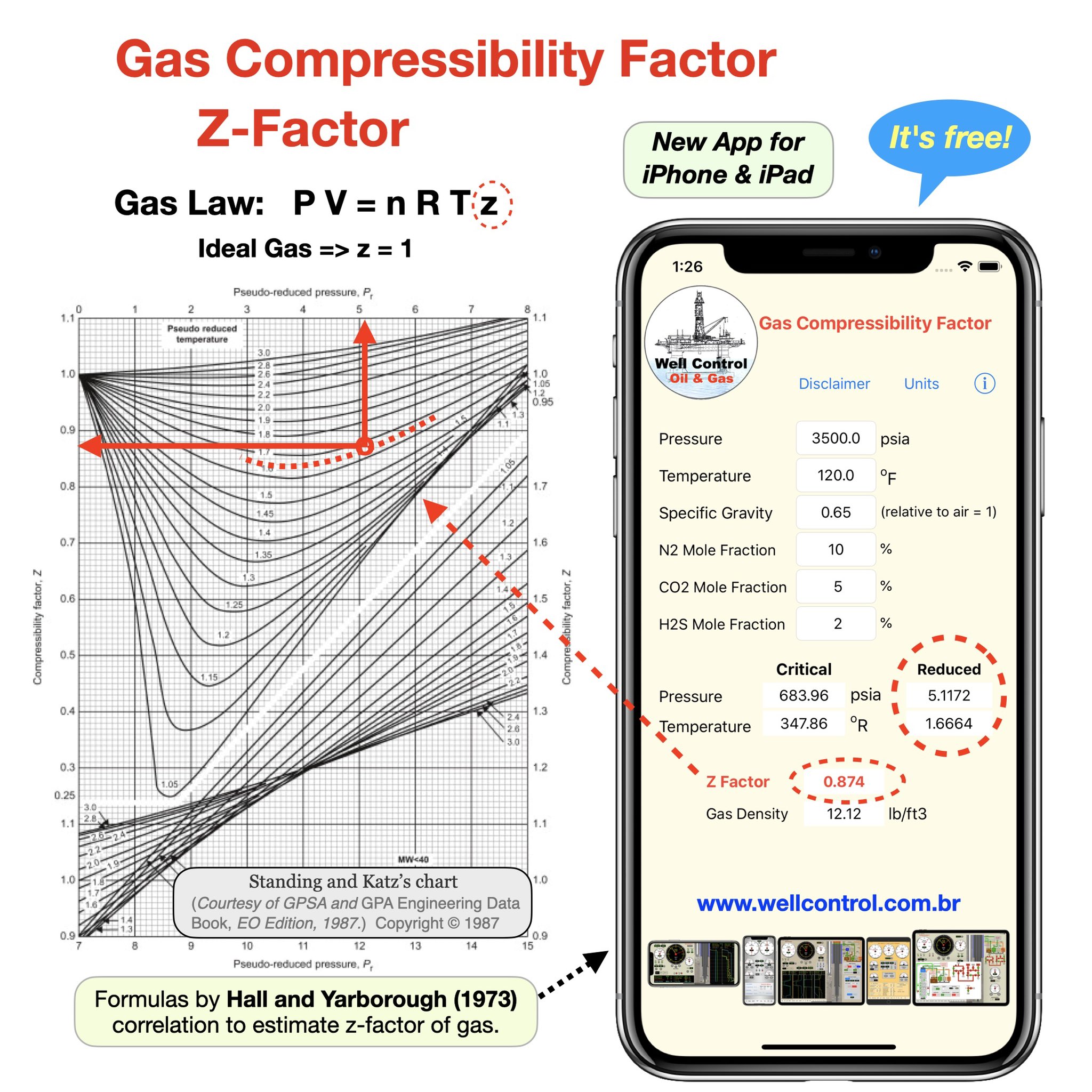
The compressibility factor of a gas is defined as $Z = pV/(nRT)$. If attractive intermolecular forces dominate then $Z$ tends to be smaller than 1, and vice versa if repulsive forces dominate. In
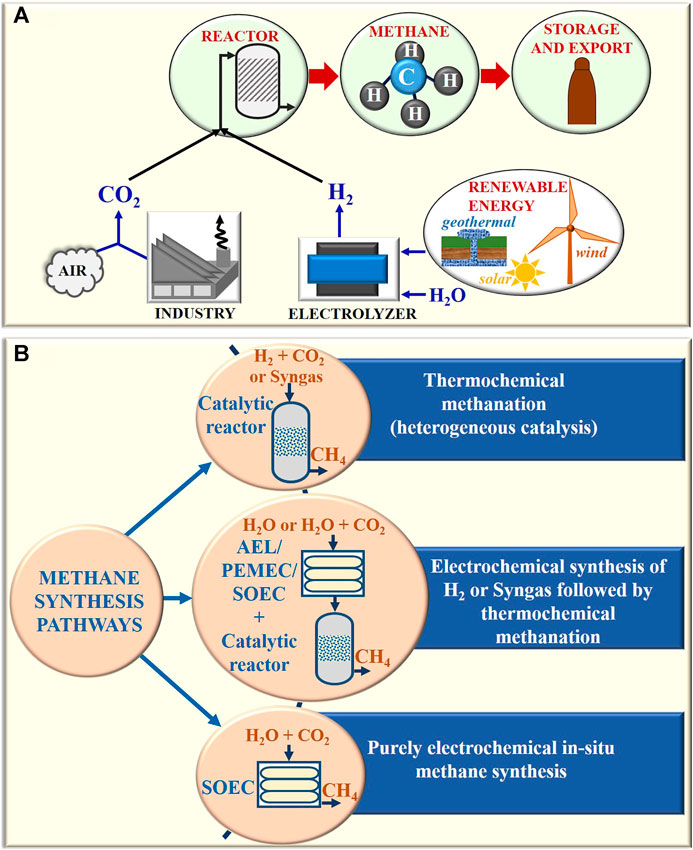
Frontiers A Review on Synthesis of Methane as a Pathway for

Net-zero emissions chemical industry in a world of limited
Fuels, Free Full-Text

Review of the Decomposition of Ammonia to Generate Hydrogen
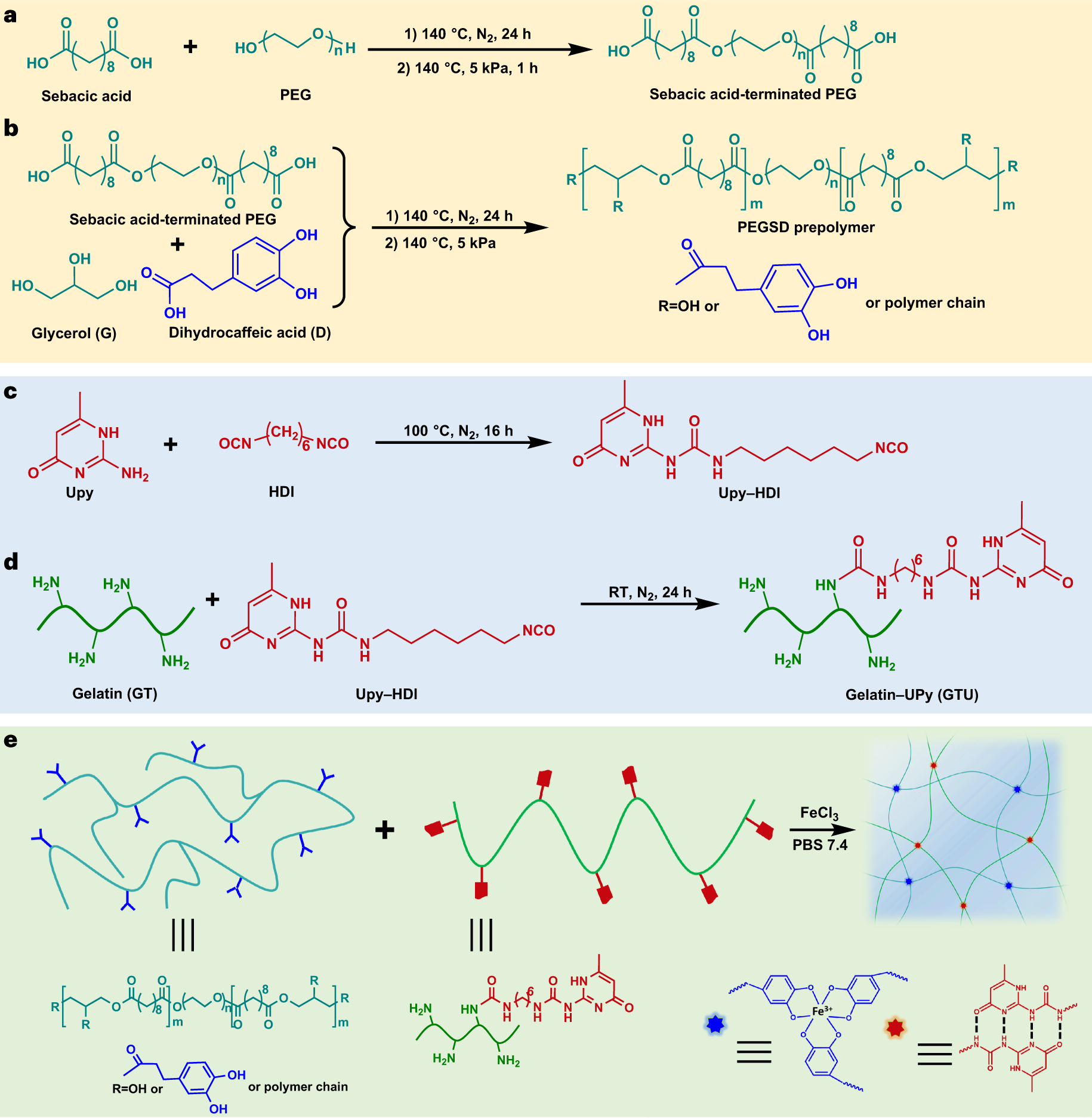
Physical dynamic double-network hydrogels as dressings to
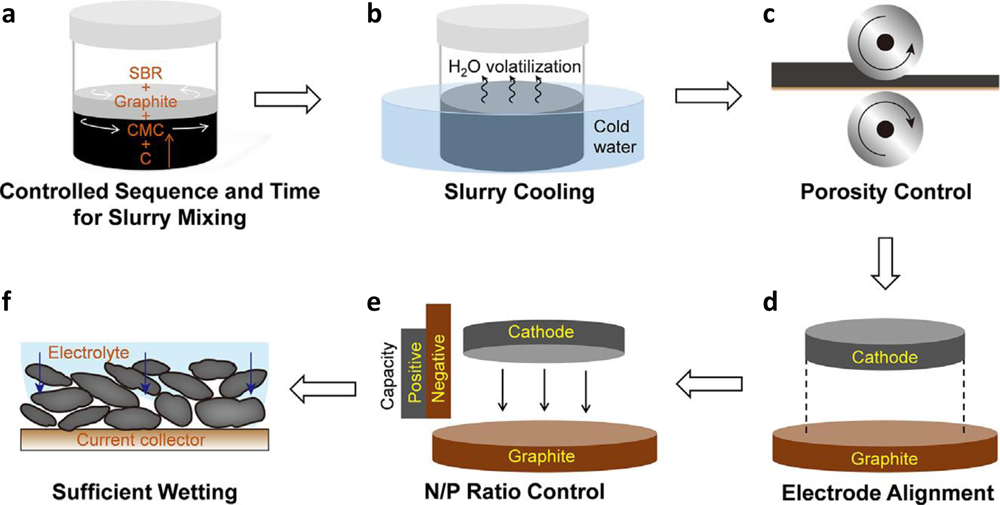
Best practices in lithium battery cell preparation and evaluation
Why compressibility factor of areal gas is greater than unity at

Effects of mechanical pressure on anion exchange membrane water
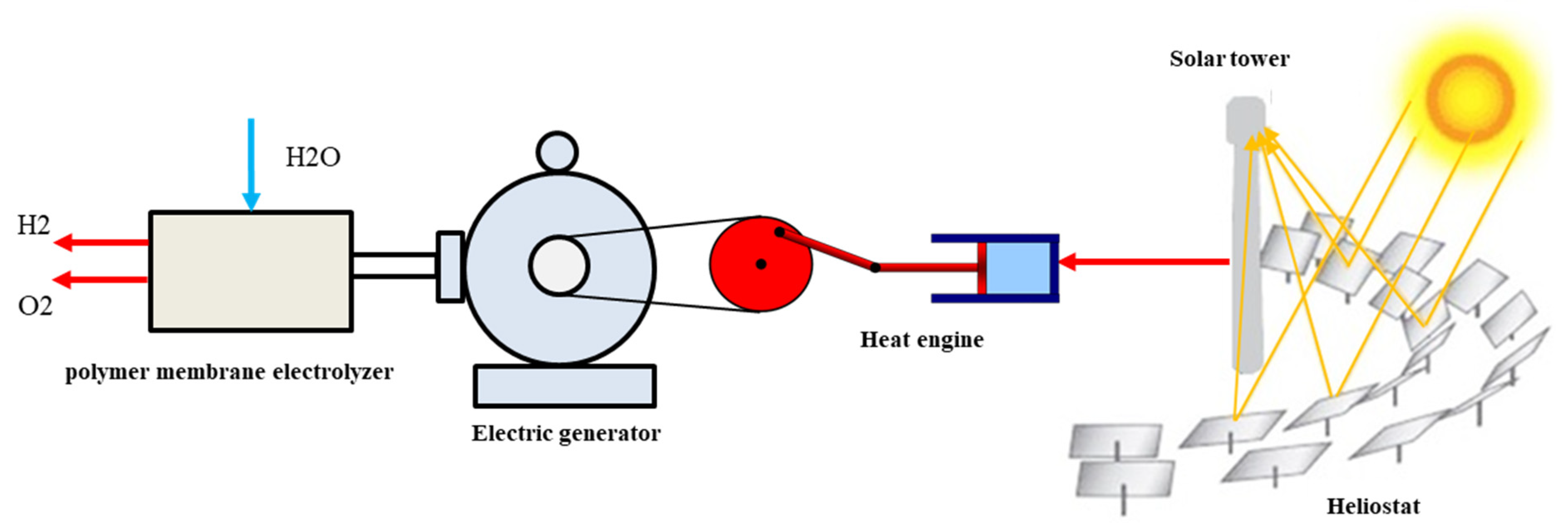
Molecules, Free Full-Text

Heat pump - Wikipedia
Related products