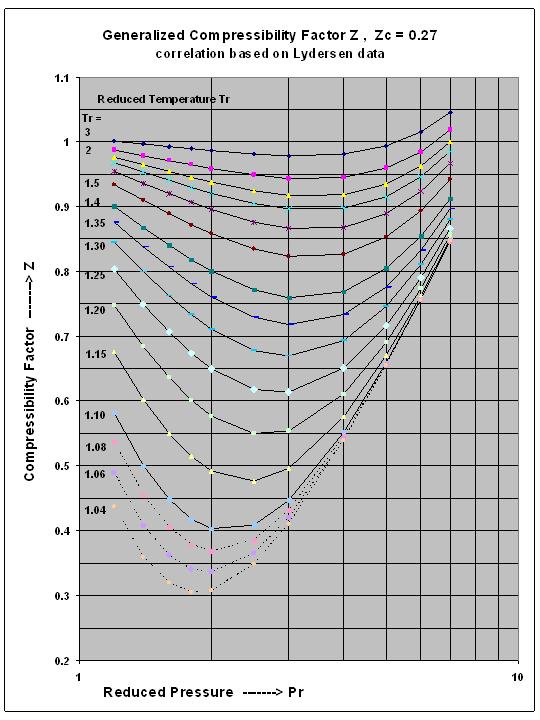
Compressibility factor Z = PV / nRT is plotted against pressure as
Compressibility factor Z = PV / nRT is plotted against pressure as shown below:What is the correct order for the liquefiability of the gases shown in the above graph? A. CO 2
Compressibility factor Z - PV - nRT is plotted against pressure as shown below-What is the correct order for the liquefiability of the gases shown in the above graph- A- CO 2- CH 4- N 2- H 2B- H 2- CH 4- N 2- CO 2C- CH 4- H 2- N 2- CO 2D- H 2- N 2- CH 4- CO 2

Compressibility factor (Z=(PV)/(nRT)) is plotted against pressure

Non-Ideal Gas Behavior Chemistry: Atoms First

PV Compressibility factor Z= nRT is plotted against pressure : N. Ideal gas What is the correct order of liquefiability of the gases shown in the above graph? H

PVCompressibility factor Z=-isnRTplotted against pressure: What isthe correct order of liquefiabilityof the
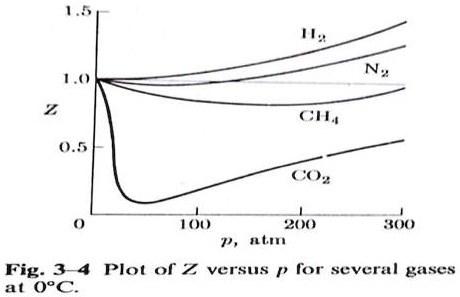
SOLVED: Subject: Compressibility Factor What is the analysis of the graph? 1.5 N 1.0 Z CHA 0.5 CO2 1 0 100 200 P, atm 300 Fig. 3-4: Plot of Z versus p for several gases at oc.
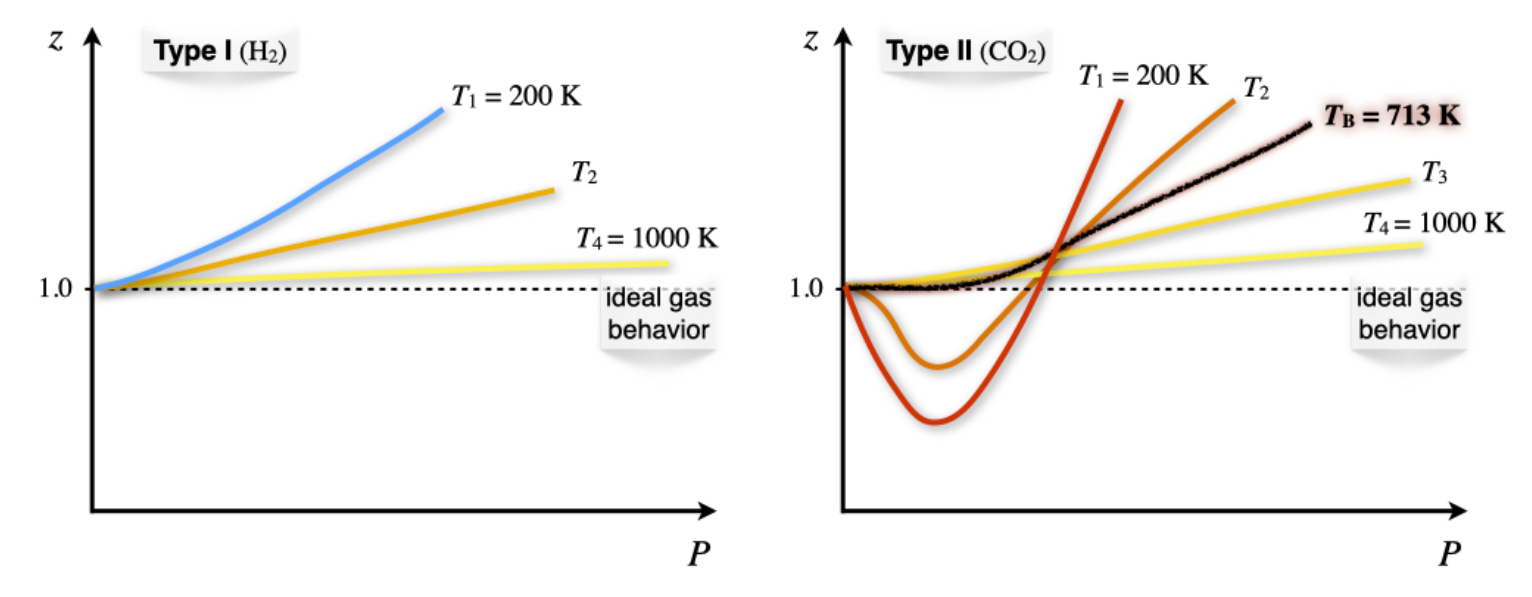
11.3: Critical Phenomena - Chemistry LibreTexts

Deviation Of Real Gas From Ideal Gas Behavior
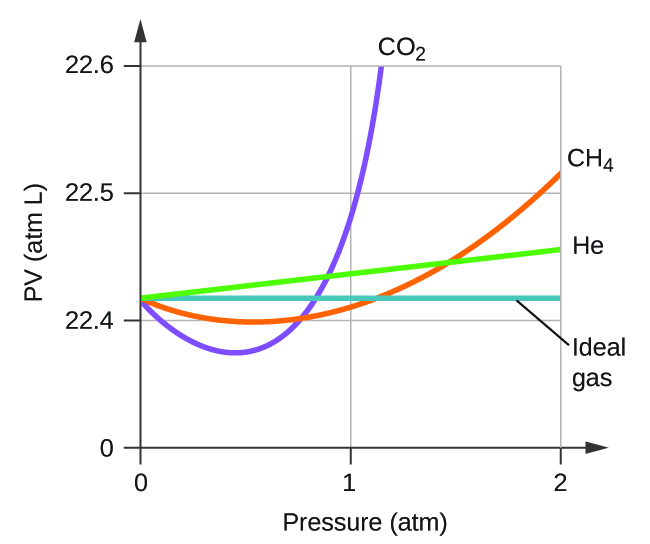
Non-Ideal Gas Behavior – Chemistry
Where is the deviation of a gas from ideal behaviour maximum at? - Quora
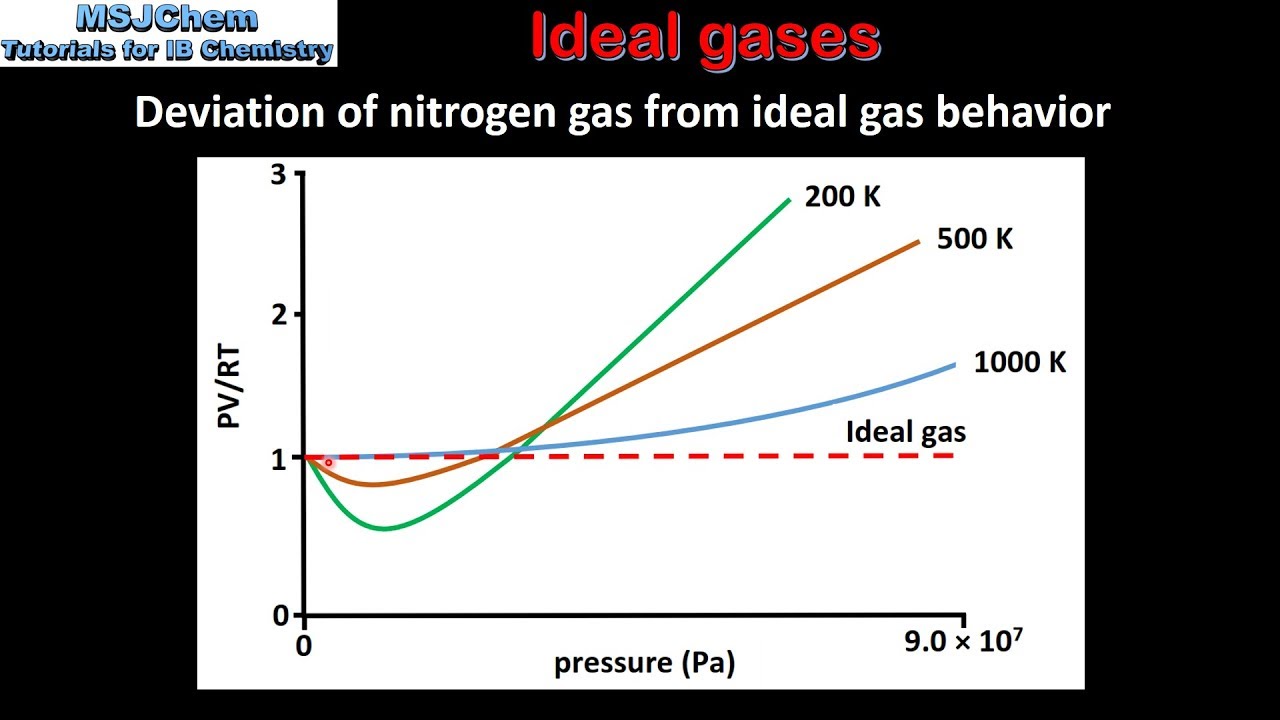
1.3 Deviation from ideal gas behaviour