What is the shape of the carbonate ion, (CO3)^2 ?
4.6
(418)
Write Review
More
$ 20.99
In stock
Description
qph.cf2.quoracdn.net/main-qimg-7fd28dcee3faa6ccf6d

Hybridization as a Way of Explaining VSEPR Theory - ppt download
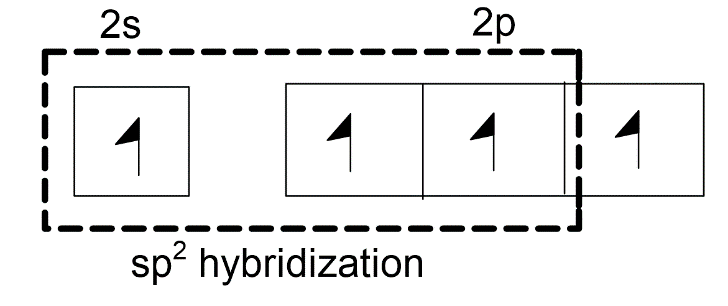
Explain hybridization of central atom in: $CO_{3}^{2-}$

Chemistry!!! Not Mystery : Delocalisation of pi π bond
Solved] Chem help. For CO32, carbonate ion, draw the Lewis structure (by

CO3 2- (Carbonate Ion) Lewis Structure

Draw the structure of CO32-. Include all lone pairs of electrons and formal charges. Draw the ion by placing atoms on the grid and connecting them with bonds. Include all lone pairs

10.3: VSEPR Geometry - Chemistry LibreTexts

Lewis Structure for CO3 2- (Carbonate ion)
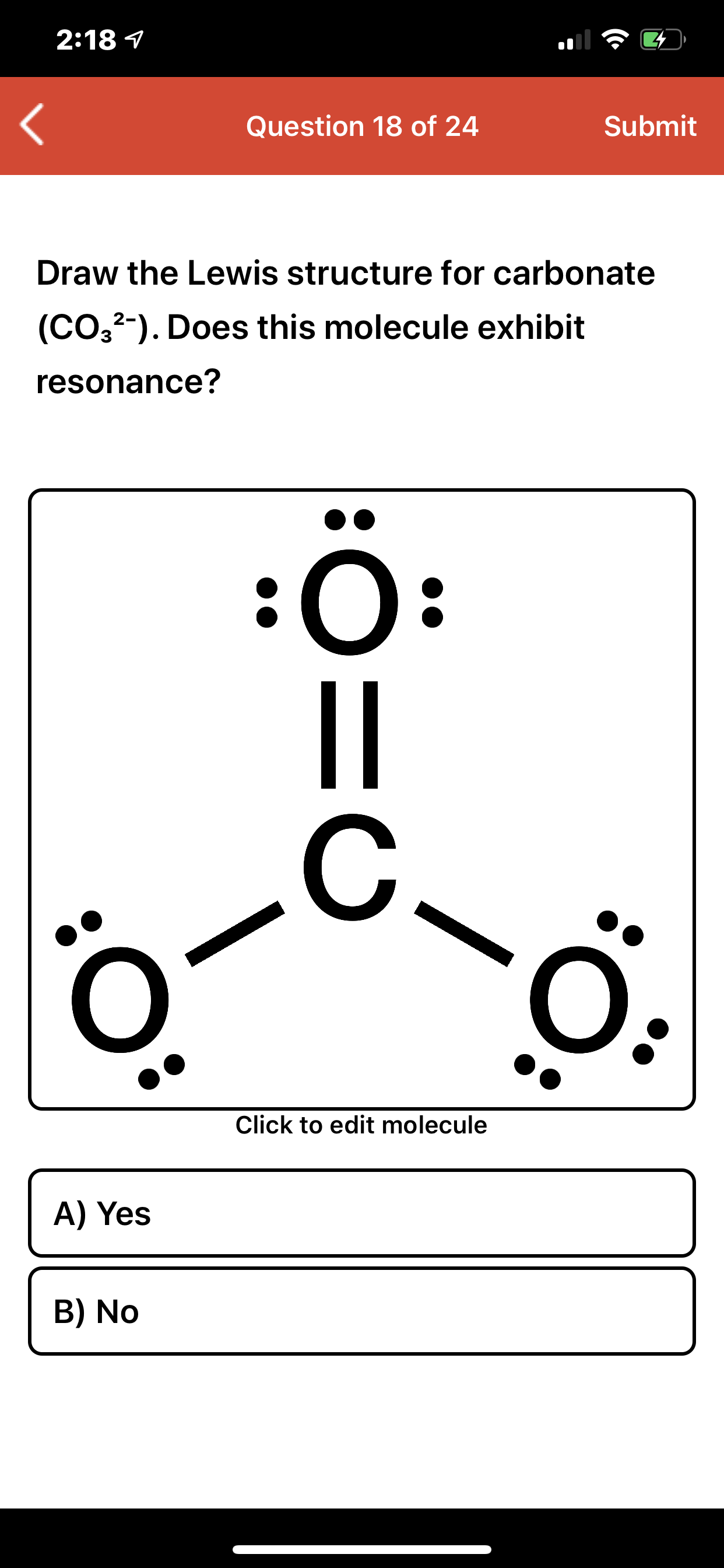
Answered: 2:18 1 Question 18 of 24 Submit Draw…

Valence bond structure of carbonate ion (CO3 2-).
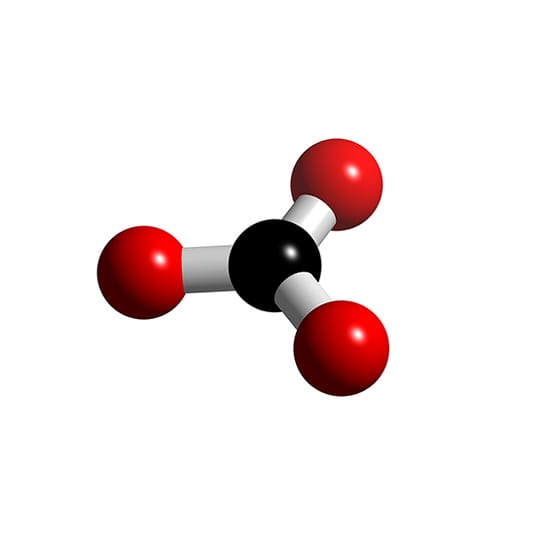
CO3]2- - Carbonate
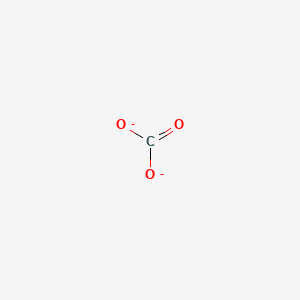
Carbonate Ion, CO3-2
Related products
You may also like