
At high pressure, the compressibility factor 'Z' is equal toa

NEET Chemistry Chapter Wise Mock Test - Mock Test 2 - CBSE Tuts

Gas Compressibility - an overview

The given graph represents the variation of Z (compressibility factor = \[\dfrac{{PV}}{{nRT}}\] ) versus P, for three real gases A, B and C. Identify the only incorrect statement.
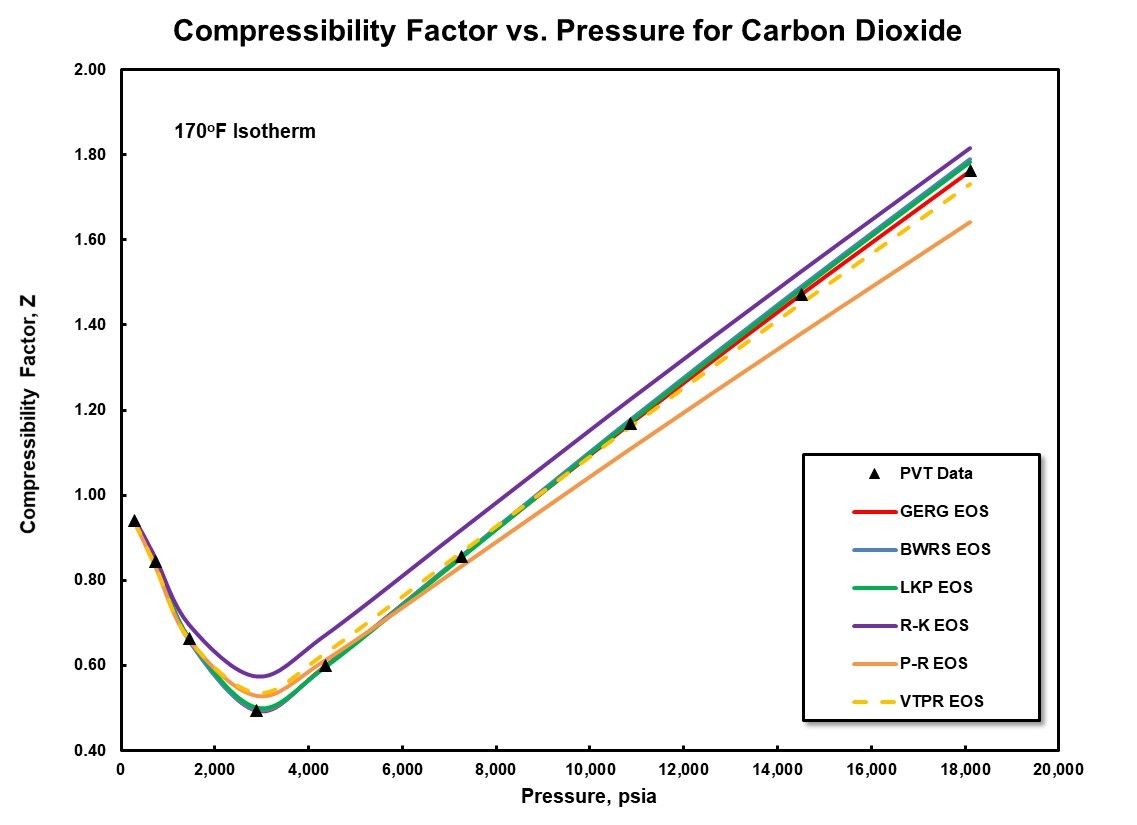
Compressor performance and thermodynamics
The compressibility factor (Z) for a real gas under high pressure is
What is compressibility factor? - Quora

The role of the compressibility factor Z in describing the volumetric behavior of gases

SOLVED: The compressibility factor, z, is used for predicting the behavior of non-ideal gases. How is the compressibility factor defined relative to an ideal gas? (Subscript c refers to critical value.) a) Z = p/pe b) Z = pV/RT c) Z = T/Tc d

For H(2) gas, the compressibility factor,Z = PV //n RT is
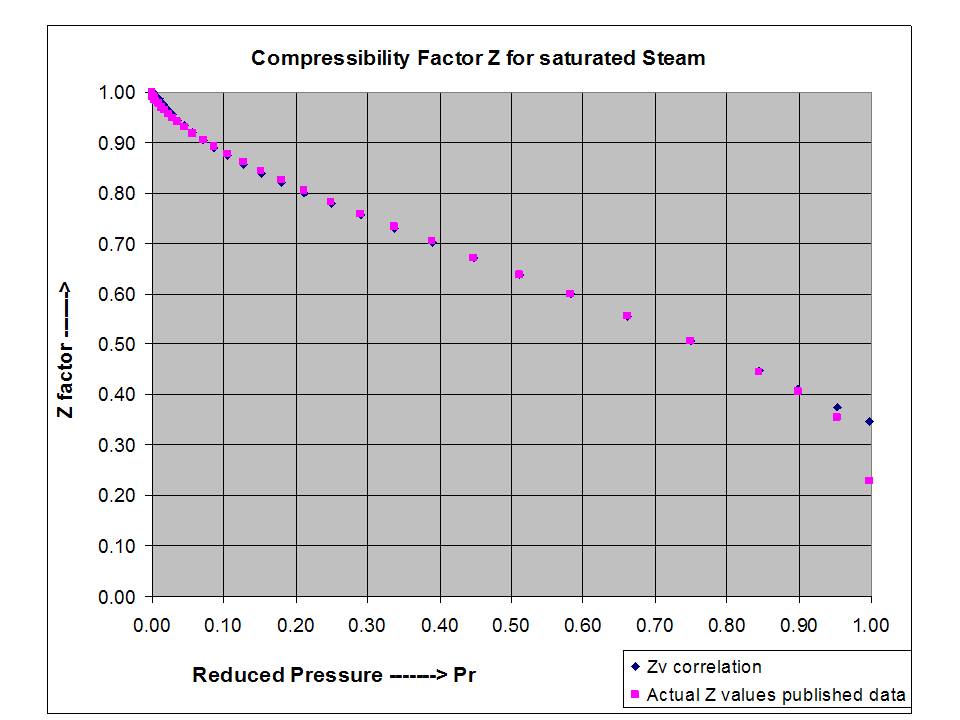
Compressibility factor Z

Compressibility Factor Mini Q bank - States of Matter States of

Boyle's temperature or Boyle point is the temperature which a real gas starts behaving like an ideal gas over a particular range of pressure. A graph is plotted between compressibility factor Z

Objectives_template

NEET Chemistry Chapter Wise Mock Test - Mock Test 2 - CBSE Tuts









