Write the expression for the compressibility factor (Z) for one mole of a gas. Write the value of Z for an

The compressiblity factor Z for 1 mole of a real gas at low pressure can be written as

Deviation of Real Gases from Ideal Gas Behaviour - GeeksforGeeks

daniloteste by Danilo Matteus - Issuu

Solved We begin by showing that the compressibility factor

Z for ideal gas is..
The compression factor (compressibility factor) for one mole of a van der Waals' gas - Sarthaks eConnect

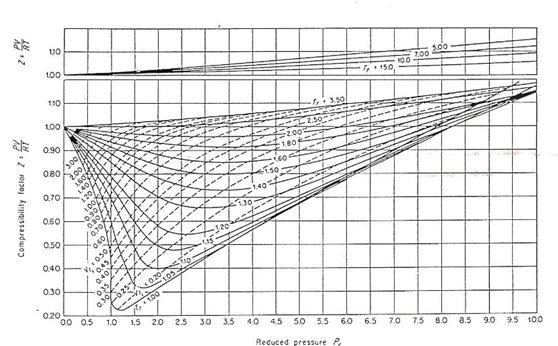
Real-gas z-factor, as attributed to Standing and Katz, 9 plotted as a

Gases

Compressibility Factor of Gas, Overview, Equation & Chart - Lesson

SOLVED: 4.17 The non-ideality of a gas may be expressed as a compressibility factor, z: PVm RT a. Find the value of z for the ideal gas. b. Given the van der

Ideal gas law - Wikiwand
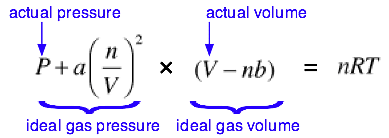
Derivation of Van Der Waals Equation: Real & One Mole of Gas