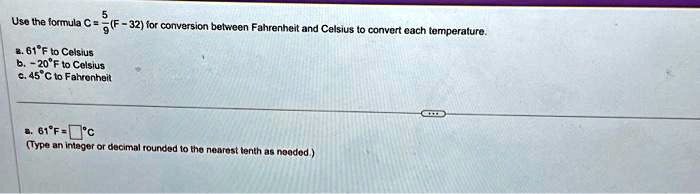
1 ltr of water at 0°c is mixed with 1 litre of water at 45°c. What's the Final temperature of water? - Quora
If 10g of ice at 0 C is mixed with 10g of water at 40 C, what is the final mass of water in the mixture? - Quora

SOLVED: 1 kg of water at a temperature of 45 °C is mixed with 1.5 kg of alcohol at 20 °C. Find the final temperature of the mixture. Take the specific heat
How much heat energy is required to convert 100 g of ice at 0 degrees Celsius into water at 50 degrees Celsius? - Quora
Why does salt lower the freezing point of water? - Quora
At what temperature will cold water become unbearable to drink? - Quora
2 kg of ice at 0°c is mixed with 8 kg of water at 20°c. What is the final temperature? - Quora

1 ltr of water at 0°c is mixed with 1 litre of water at 45°c. What's the Final temperature of water? - Quora
1 ltr of water at 0°c is mixed with 1 litre of water at 45°c. What's the Final temperature of water? - Quora
A container of water at 30.0°C experiences a volumetric change of 5.00 x 10-3 L. If the original volume of water was 425 mL, what temperature change did the water go through? - Quora
Is 1000ml of water equal to 1000g of water? - Quora
If I mix 50°C water with 10°C water, what is the temperature of the water? - Quora