
Establishing expiry date for clinical diagnostic reagents
Product shelf life is an essential product performance requirement that, along with other design requirements, is used to determine the safety and efficacy of a clinical diagnostic

January 2022 Medical Laboratory Observer

Food Expiration Date Guidelines Plus Easy-to-Read Chart

Latest News – iHealth Labs Inc

SEC Filing Akoya Biosciences, Inc.
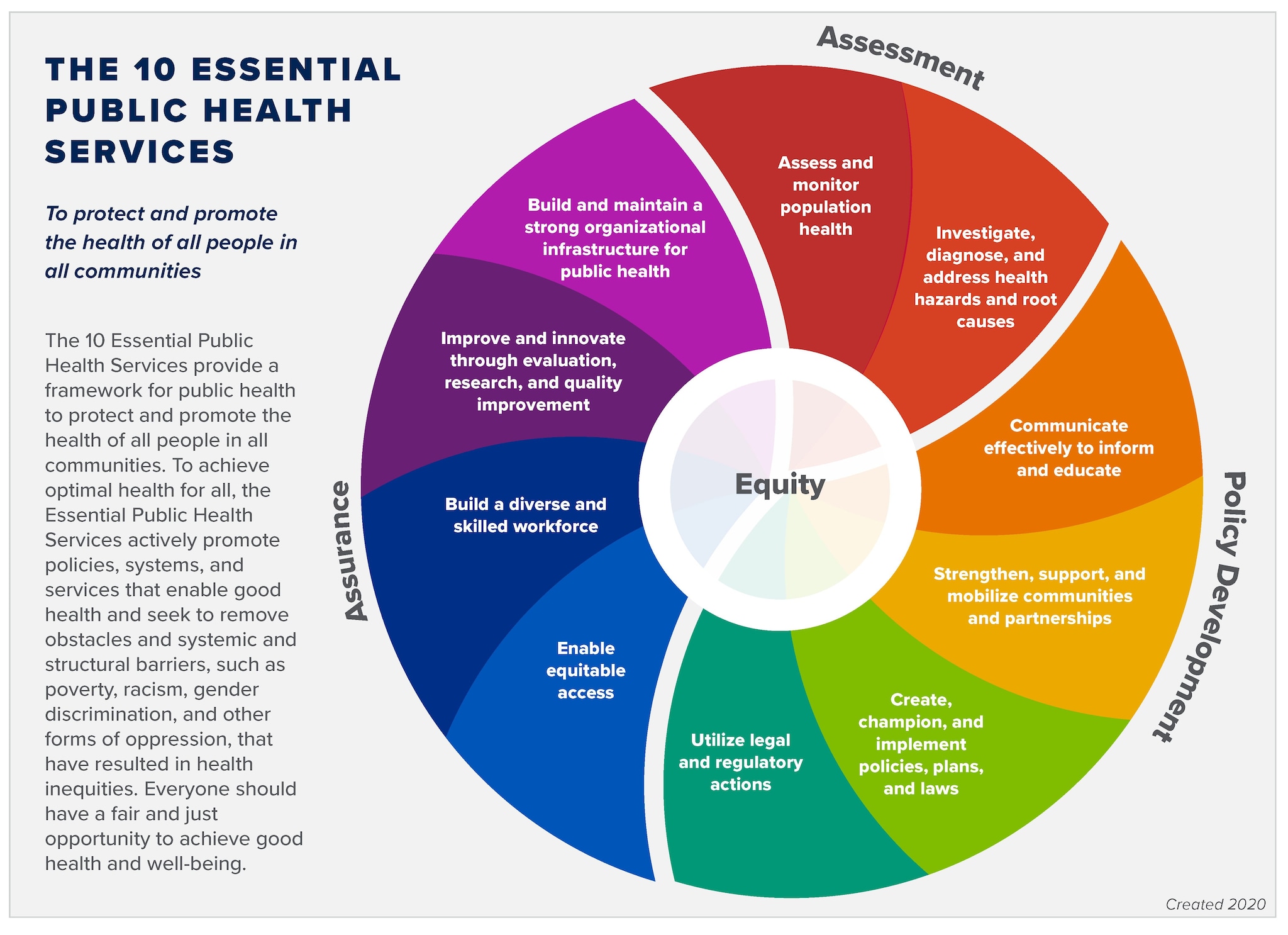
CDC - 10 Essential Public Health Services - Public Health
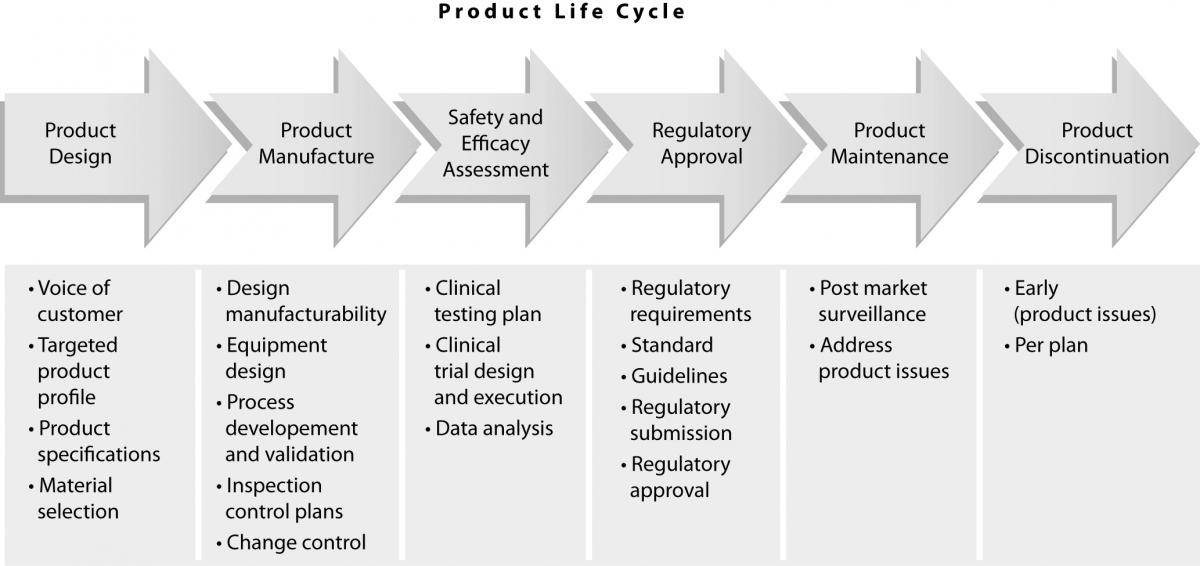
Total quality in laboratory diagnostics: the role of commercial

ASTM F2477-23 - Standard Test Methods for
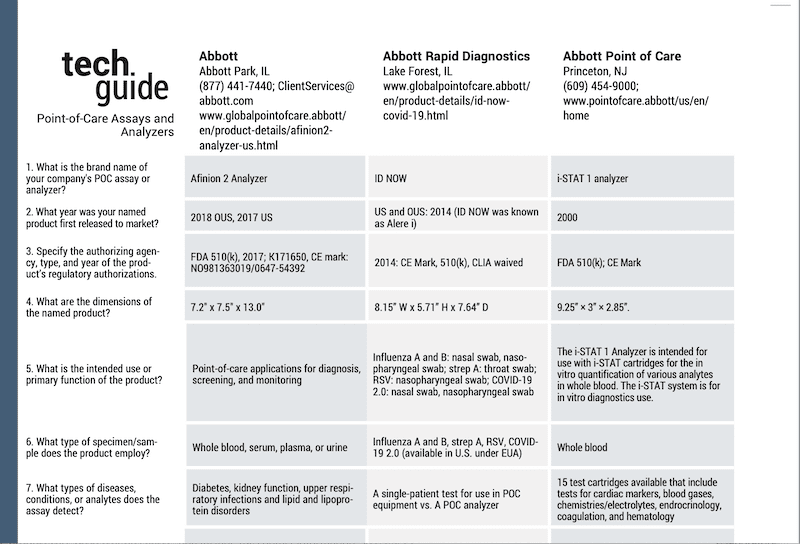
Clinical Lab Products

Shelf life - Wikipedia
National Action Plan to Build Australia's Diagnostic Technology

Update: UN Day Ministry of Health

What are Nutraceuticals?

Autoverification of test results in the core clinical laboratory

Phases in the development of plant clinics and the Plant Health

How to find out your eyesight prescription: Where and what to know








