The compression factor (compressibility factor) for one mole of a van der Waals' gas - Sarthaks eConnect
The compression factor (compressibility factor) for one mole of a van der Waals

physical chemistry - Is the compressibility factor smaller or greater than 1 at low temperature and high pressure? - Chemistry Stack Exchange

If `Z` is a compressibility factor, van der Waals' equation at low pressure can be written as
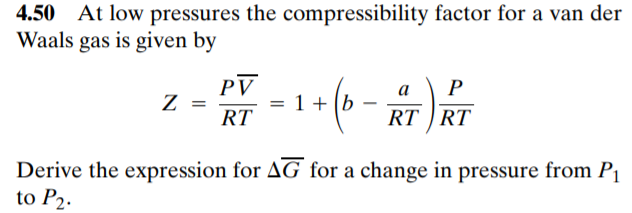
Solved 4.50 At low pressures the compressibility factor for
Using van der Waals' equation, calculate the constant 'a' when two moles of a gas confined - Sarthaks eConnect

States Of Matter Notes: Class 11, JEE, NEET, AIIMS
a' and 'b' are van der Waals' constants for gases. Chlorine is more easily liquefied than ethane because - Sarthaks eConnect
For one mole real gas, the correct value of Z at point P using following graph is - Sarthaks eConnect

At low pressures the compressibility factor for a van
If Z is a compressibility factor, van der Waals' equation at low pressure can be written as - Sarthaks eConnect
The compressibility factor for a real gas at high pressure is - Sarthaks eConnect

Write the expression for the compressibility factor (Z) for one mole of a gas. Write the value of Z for an

States Of Matter Notes: Class 11, JEE, NEET, AIIMS

The graph of compressibility factor (Z) v/s P 1 mol of a real gas is shown in following diagram. The graph is plotted 273 K temperature. If slope of graph very high

States Of Matter Notes: Class 11, JEE, NEET, AIIMS

The compression factor compressibility factor for 1 mole of a van der Waals' gas at 0∘ C and 100 atmospheric pressure is found to be 0.5 . Assuming that the volume of






