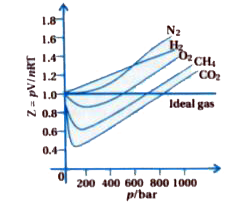
In the following compressibility factor Z vs pressure graph at 300 K, the compressibility of CH 4 at pressure
In the following compressibility factor Z vs pressure graph at 300 K, the compressibility of CH 4 at pressure
In the following compressibility factor Z vs pressure graph at 300 K- the compressibility of CH 4 at pressure -200 bar deviates from ideal behaviourA- The molar volume of CH 4 is less than its molar volume in the ideal stateB- The molar volume of CH 4 is same as that in its ideal stateC- Intermolecular interactions between CH 4 molecules decresasesD- The molar volume of CH 4 is more than its molar volume in the ideal state
Solved The first order rate constant of the decomposition of

KVPY-SX 2016 Chemistry Question Paper with Solutions PDF Download
The graph of compressibility factor (Z) vs. P for one mole of a

47. In the following compressibility factor (Z) vs pressure graph 300 K, the compressibility factor of CH4 pressures < 200 bar deviates from ideal behavior because
Gujrati] Explain compressibility factor (Z).

KVPY-SX 2016 Chemistry Question Paper with Solutions PDF Download

Chemical Thermodynamics

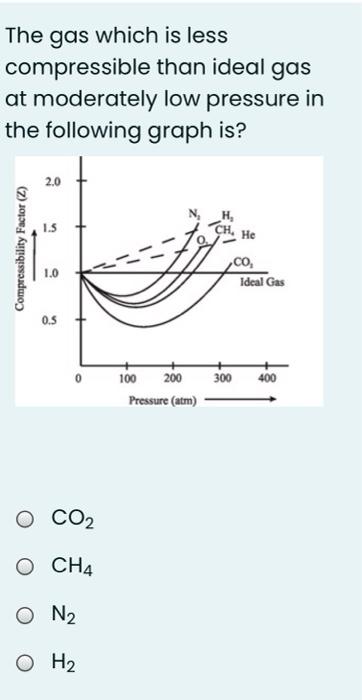
physical chemistry - Compressibility Factor Graph - Which gas
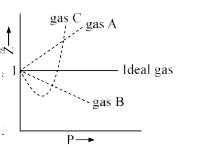
Gas C is a real gas and we can find 'a' and 'b' if intersection data i

KVPY-SX 2016 Chemistry Question Paper with Solutions PDF Download

Real Gas: Difference, Examples and Factors

The graph of compressibility factor (Z) vs. P for one mole of a real g

Punjabi] The graph of compressibility factor (Z) vs. P for one mole o