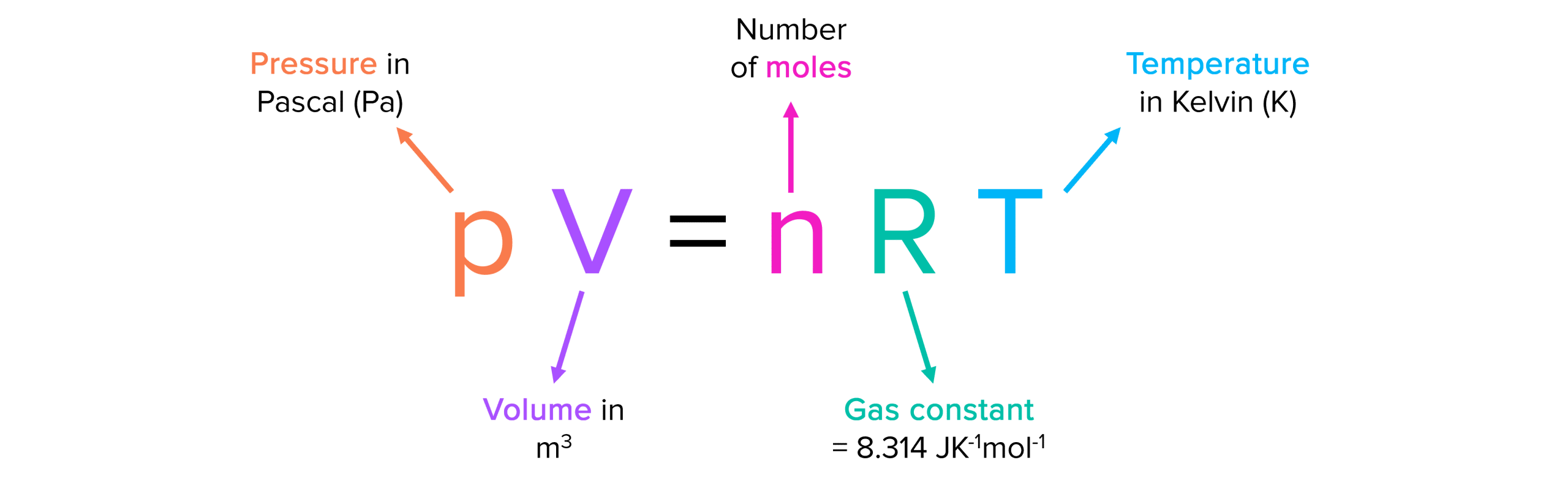Kinetic Theory of an Ideal Gas: Equation, Assumption, Concept, Examples
4.9
(299)
Write Review
More
$ 5.50
In stock
Description
Can you imagine a collection of spheres that collide with each other but they do not interact which each other. Here the internal energy is the kinetic energy. In this article, we shall understand what kinetic theory of an ideal gas is.

Kinetic Theory: Atomic and Molecular Explanation of Pressure and
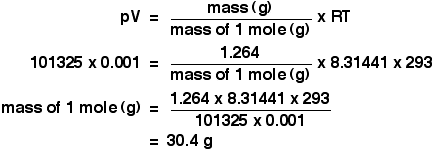
Ideal gases and the ideal gas law: pV = nRT
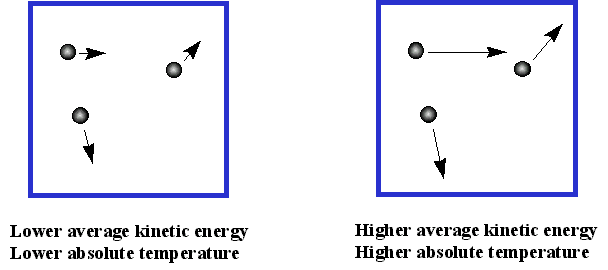
Basics of Kinetic Molecular Theory - Chemistry LibreTexts
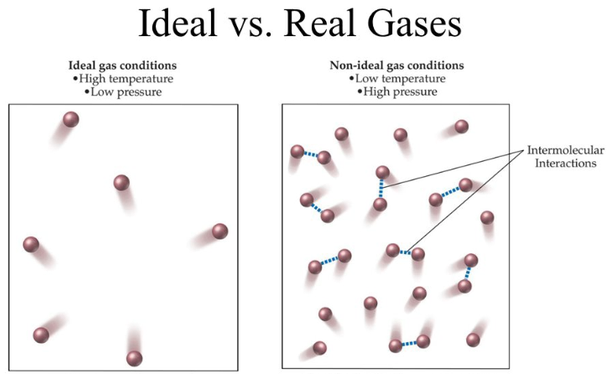
Ideal gas approximation - Energy Education

Kinetic Theory Of Gases - Assumptions, Derivation, Formula
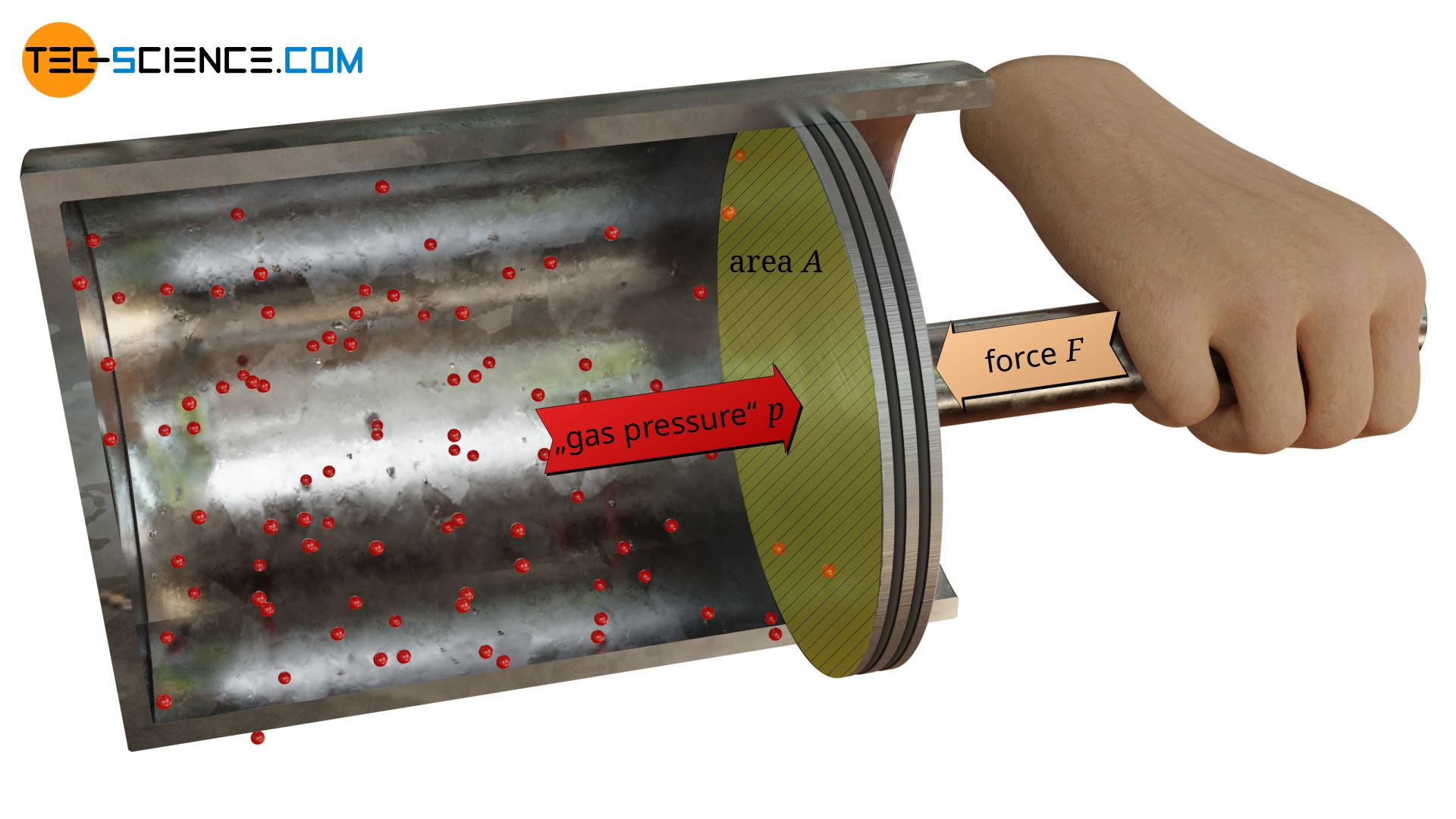
Pressure and temperature (kinetic theory of gases) - tec-science
CBSE Class 11 Physics Kinetic Theory Of Gases Notes

Kinetic Molecular Theory - Video Tutorials & Practice Problems

Lecture 1 the kinetic theory of gases
You may also like