
The compression factor (Z) Co, 7°C and 100 atm is 0.21. Calculate
Click here:point_up_2:to get an answer to your question :writing_hand:the compression factor z for co at 7c and 100atm is 021 calculate the volume
Click here👆to get an answer to your question ✍️ The compression factor -Z- Co- 7-C and 100 atm is 0-21- Calculate the volume of a 4 mole sample of co- same temperature and pressure -use R - 0-08 L- atm-K-mol -1- 0-192 -2- 0-05 L -3- 0-38 L -4- 0-44 L closed container can be

Compression Factor Exam Problem using Molar Volumes - Fully

5 Common Functional Implied Volatility Analysis - Statistik - HU

Chemical Process Engineering - Harry Silla - Ventech!
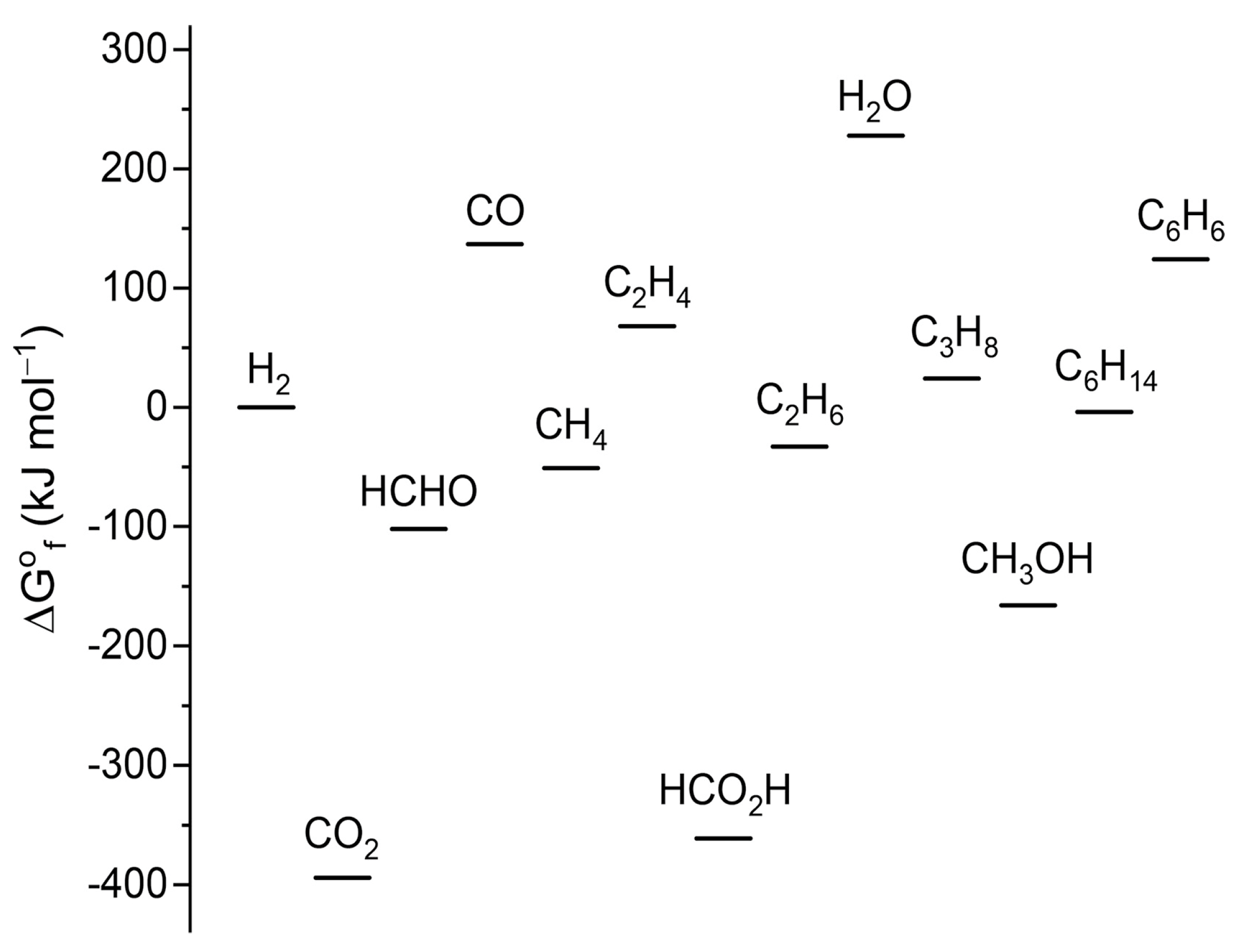
Inorganics, Free Full-Text

PDF) Simple methods for the calculation of thermodynamic

Processes, Free Full-Text

The compressibility factor for a given real gas is 0.927 at 273 K and

Solved The compression factor, Z, can also be calculated
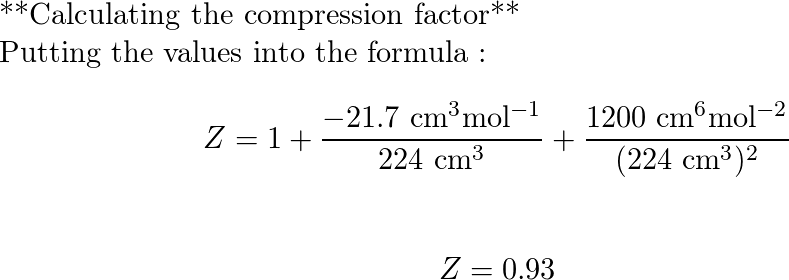
At 273 K measurements on argon gave B = -21.7 cm$^3$ mol$^{

Solved 1) The compression factor, Z, can be written as: Z

Ch.13



:max_bytes(150000):strip_icc()/Term-Definitions_zscore.aspsketch-3c68631c0479475f9bee1d6ecb0f4e23.jpg)





