
Ideal gas law, Definition, Formula, & Facts
4.8
(441)
Write Review
More
$ 20.99
In stock
Description
Ideal gas law, relation between the pressure P, volume V, and temperature T of a gas in the limit of low pressures and high temperatures, such that the molecules of the gas move almost independently of each other. In such a case, all gases obey an equation of state known as the ideal gas law: PV =
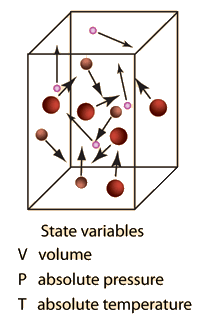
Ideal Gas Law

Boyle's law - Wikipedia

Difference Between Ideal Gas Law and Real Gas Law

Combined Gas Law, Definition, Formula & Example - Lesson

Using the Equation of State for an Ideal Gas, Chemistry

i1.wp.com//wp-content/upl
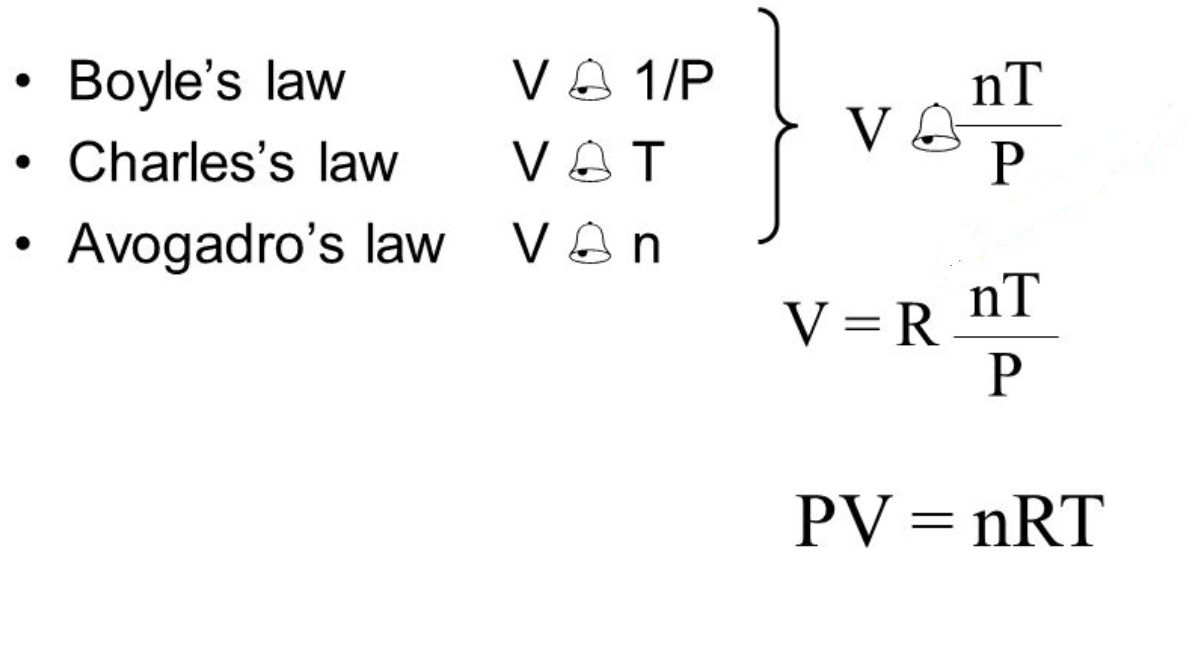
Gas Laws

The Ideal Gas Law, Equation & Constant

Ideal Gas Constant (R) - Universal Gas Constant

01 part1-ideal-gas

Calculating Pressure of an Ideal Gas - Derivation
How to calculate the characteristic gas constant of a gas (air
You may also like









