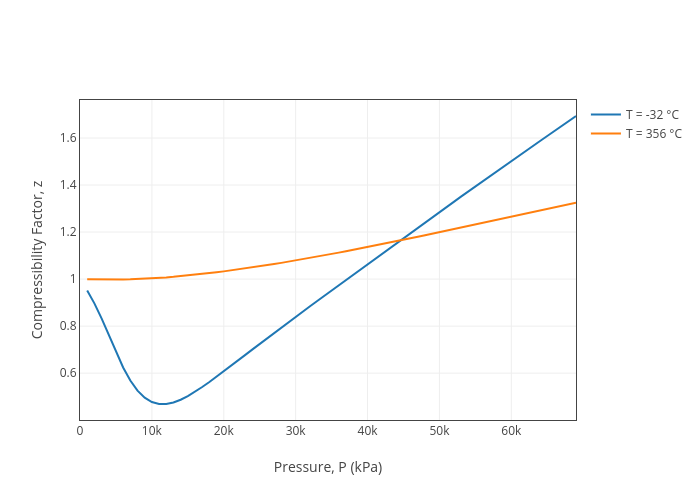
Compressibility Factor, z vs Pressure, P (kPa)
Jdvani's interactive graph and data of "Compressibility Factor, z vs Pressure, P (kPa)" is a line chart, showing T = -32 °C vs T = 356 °C; with Pressure, P (kPa) in the x-axis and Compressibility Factor, z in the y-axis..

Superheated water vapor is at 40 Mpa, 550 degree Celsius. Can you consider this vapor as an ideal gas? Support your answer by calculating the compressibility factor. a) What is the specific

SOLUTION: Thermo - Studypool

plotting - How to plot Compressibility factor Z vs Pressure P using ParametricPlot? - Mathematica Stack Exchange

The graph of compressibility factor Z vs P for one mole of a real gas is shown in following diagram. The graph is plotted at a constant temperature 273 K. If the

Solved QUESTION 4 Determine the reduced temperature TR for
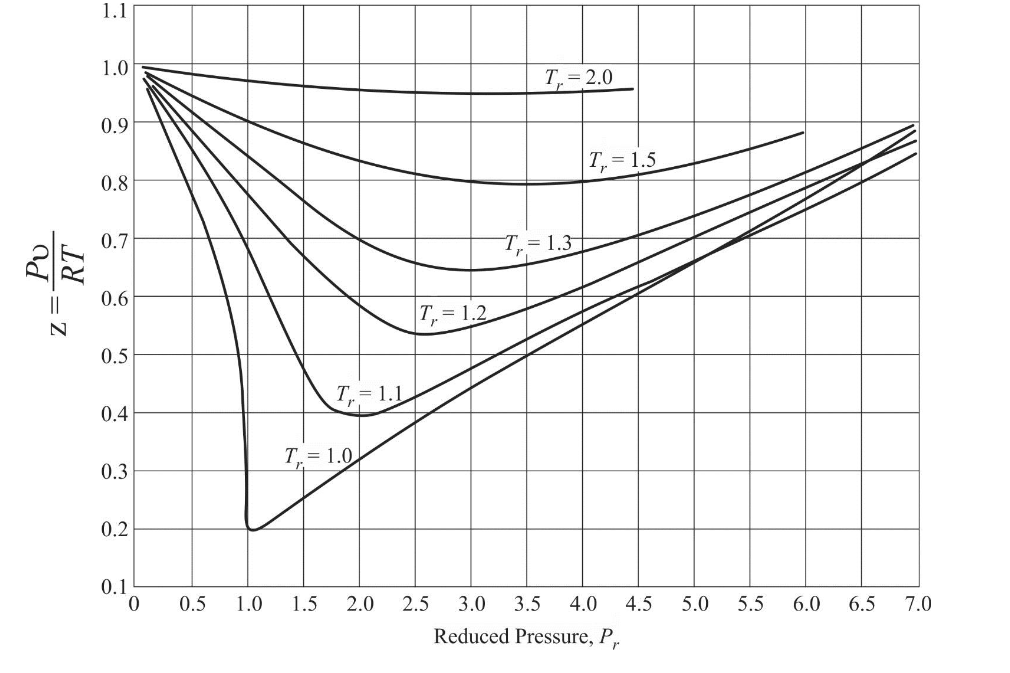
Solved Problem 8 (25 points) p 10,060 kPa The pressure gage
Is z (compressibility factor) vs P (pressure) graph drawn by changing volume? If it is why it isn't drawn by changing mole - Quora

Short handy Formulas calculating Densities and Enthalpies of Saturated Steam

Variation of pseudo-compressibility Z factor vs. pressure and hydrogen

Maximum pressure (P max ) (in kPa) generated for each measure according

plotting - How to plot Compressibility factor Z vs Pressure P using ParametricPlot? - Mathematica Stack Exchange

Gas compressibility factor Z: Ideal gas vs Real gas